
Success story second part Solothurn site, GMP news & insight into the lab
Dear Cannerald and CannerGrow Community,
in the last blog article (click), you got a detailed insight into the development of our site in Fraubrunnen.
Today we dedicate ourselves to the location in Solothurn (2nd production facility). We will give you an intensive insight into the topics of GMP certification and the development and expansion of our laboratory area. We will give you exciting information and furthermore we will give you an update about the current time windows.
Our second location in Solothurn
After you have literally snatched the plants of our first location in Fraubrunnen out of our hands at the beginning of December, we started the sale in the 2nd hall in Solothurn on 12.12.2021. At the start of the sale, the hall was still completely empty. In the following teaser, you can see what the hall looked like at the start of the sale.
Construction work started here as early as the end of 2021 and today the hall already looks very different. In November 2020, we inspected the second location for the first time. At that time, we already started planning for the complete expansion of the new production facility. Demeter will start as planned in the third quarter and Poseidon in the fourth quarter. We are pleased to be able to show you new impressions of the current construction progress.

The most important milestone in the company's history - GMP certification
With the achievement of the GMP certification by Swiss Medic, which is the most important milestone in our still young company history, we will have a unique selling point in the whole of Switzerland. While an already demanding ISO certification proves the conformity of a company's quality management, the GMP certification is much more demanding.
Since it will probably take some time until the THC area (cannabis with more than 1% THC) will be legalized, we have focused early on pharmaceutical production. Which now more and more turns out to be the right way.
👉 Click: Blog article about the THC exemption
The cleanroom for the GMP area was originally planned for Fraubrunnen. (see blue marked area) However, we have now decided to use an area of 1,050 m² as a cleanroom in Solothurn, which will be completed in the summer of 2022. Without equipment and machines, the costs are about 4,500 - 6,500 Euro per square meter. As soon as the areas are completed, we will inform you.
Since the Swiss authorities (Parliament and Federal Council) will open the export of medicinal cannabis this year in the summer (third quarter), so we are perfectly on schedule.
Picture 1: Planned clean room Fraubrunnen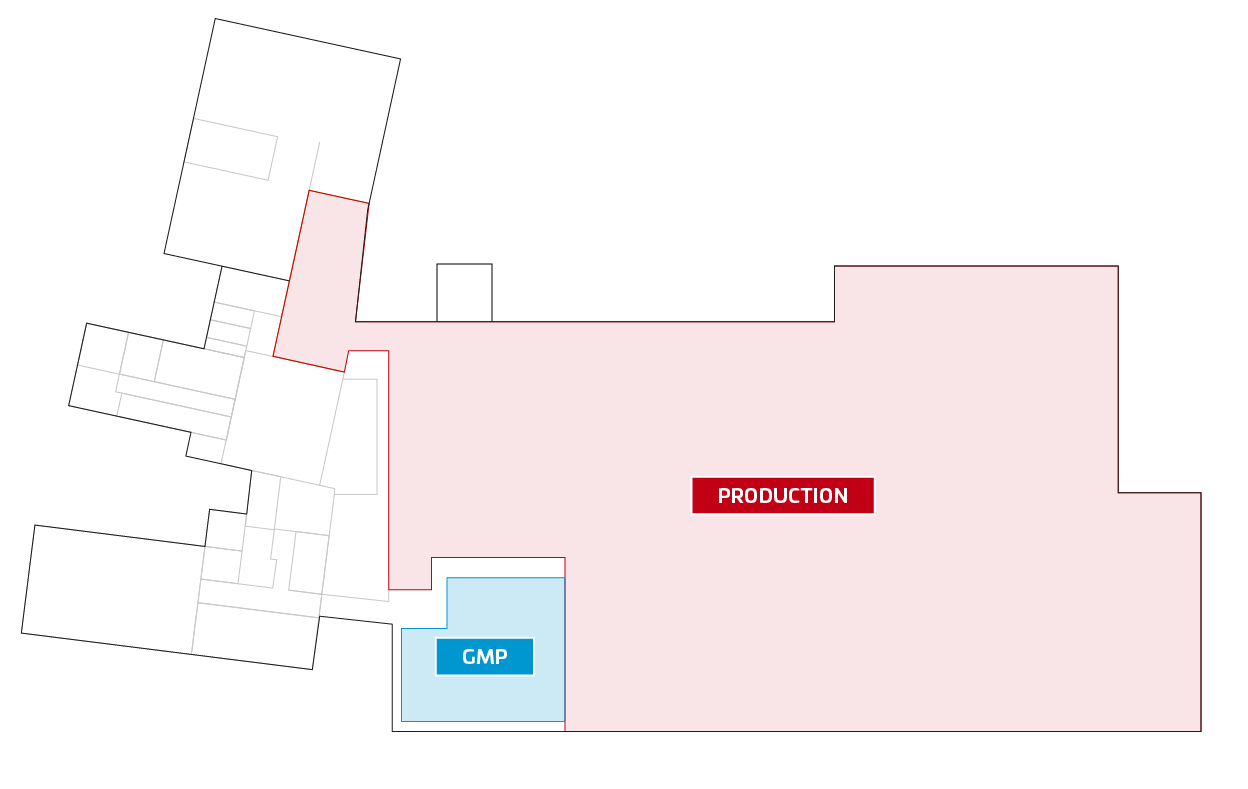 Picture 2: New planned cleanroom in Solothurn
Picture 2: New planned cleanroom in Solothurn
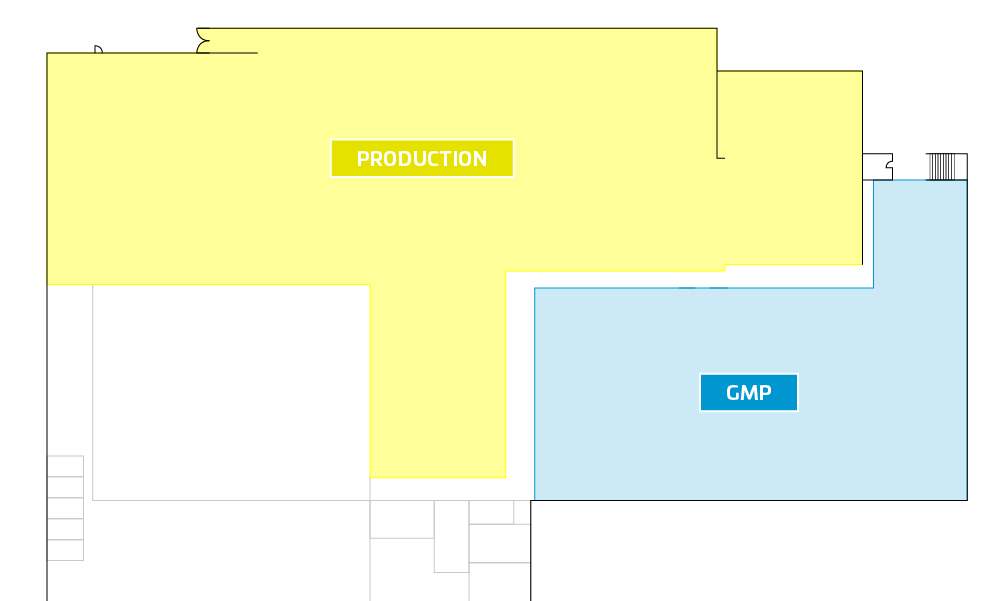
The next steps on the way to GMP certification
After the completion of the cleanroom, we will submit the remaining documents so that Swiss Medic can start the final audit. After that, we will switch production to the medical area. As the only company in Switzerland, this will open up considerable competitive advantages. When the final step has been taken, we will all be able to say that the long wait has really been worth it.
Conclusion: In addition to Fraubrunnen, the developments at the Solothurn site in 2022 are also the most important steps that the company has taken since its foundation in 2017.
Cost-efficient and future-oriented thanks to our own laboratory
Our laboratory area in Fraubrunnen will also be completed by the summer.
The head of research of our company will enormously expand the research area there together with two laboratory employees and our Research and Development team. Among other things, research projects will be carried out for the Swiss Federal Office of Public Health, in addition to various study programs in which we will participate with our partners.
The internal laboratory will allow us to approve batch releases (release of a harvest for export as a medicine) internally, which will save us a lot of time and money.
Since cannabis research is still in its infancy, we are very confident about the future! Even though we are currently running this work as a non-profit project, we can be sure that in the future we will have made a significant contribution to curing many people's health problems.
Our doctor and head of research will go into more detail about the different areas at a later date and will take you on a virtual tour of the then-completed laboratory area in the summer.
#WeGrowForYou
Your Cannerald and CannerGrow team 🌱